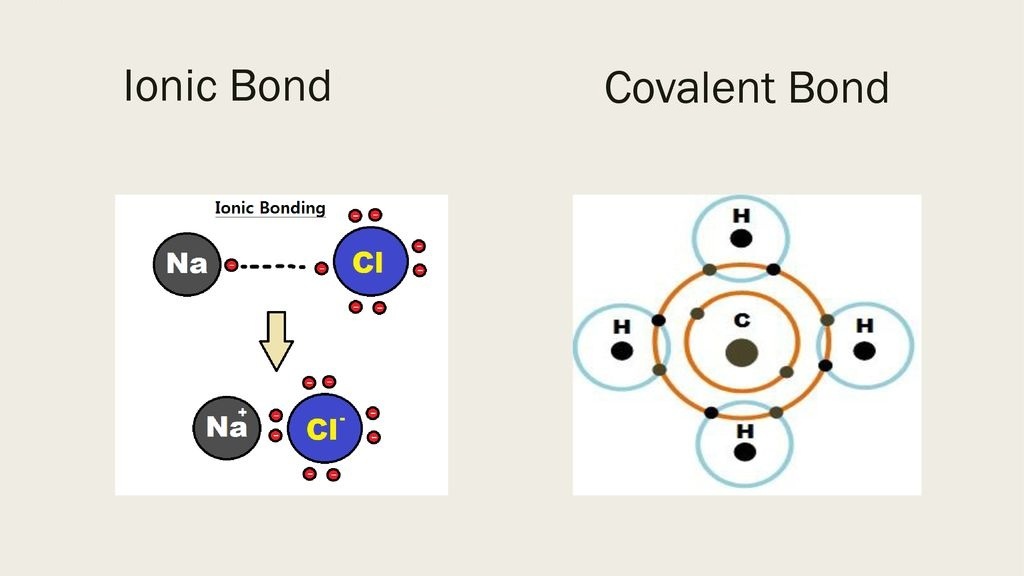
Two molecules of water can form a hydrogen bond between them. 113 10663īalasubramanian S, Pal S and Bagchi B 2002 Phys. In a discrete water molecule, water has two hydrogen atoms and one oxygen atom. B105 11929 Xu H, Stern H A and Berne B J 2002 J. 76 928 Luzar A and Chandler D 1996 Nature ( London) 379 53 Weast R C et al (eds) 1989 CRC handbook of chemistry and physics (Florida: CRS Press, Boca Raton)Īllen M P and Tildesley D J 1987 Computer simulation of liquids (Oxford)Ĭhowdhury S and Chandra A 2006 J. Koneshan S, Rasaiah G C, Lyndel-Bell R M and Lee S H 1998 J. 111 209īerendsen H J C, Grigera J R and Straatsma T P 1987 J. B110 6332ĭu H, Rasaiah J C and Miller J D 2007 J. Analyses of three-dimensional structures, particularly of proteins, reveal many consistent patterns, which are described in this review. Omta A W, Kropman M F, Wouterson S and Bakker H J 2003 J. Hydrogen bonds are weak non-covalent interactions, but their directional nature and the large number of hydrogen-bonding groups mean that they play a critical role in the structure and function of proteins and nucleic acids. Omta A W, Kropman M F, Wouterson S and Bakker H J 2003 Science 301 347 Bruni F, Ricci M A and Soper A K 2007 Phys. Yamaguchi T, Yamagami M, Ohzono H, Wakita H and Yamanaka K 1996 Chem. One of the atoms is hydrogen, while the other may be. 108 4039ĭe Jong P H K, Neilson G W and Bellissent-Funel M C 1996 J. A hydrogen bond is an attraction between two atoms that already participate in other chemical bonds. Wallen S L, Pfund D M and Fulton J L 1998 J. Pfund D M, Darab J G, Fulton J L and Ma Y 1994 J. Smith J D, Saykally R J and Geissler P 2007 J. Hydrogen bonding has a very important effect on the properties. A hydrogen bond is formed by a dipole-dipole force between an electronegative atom (the hydrogen. Marshall W L and Frantz J D 1980 In Thermodynamics of aqueous systems with industrial applications (ed.) S A Newman (Washington DC: American Chemical Society) vol 133, pp 569–581Īmo Y and Tominaga Y 2000 Physica A275 33 This attraction or hydrogen bond can have about 5 to 10 of the strength of a covalent bond. Structural Biochemistry/Chemical Bonding/Hydrogen bonds. 
A relationship of the ILs thermal behavior (expressed as melting and decomposition points) as a function of the internal hydrogen bonding extent is illustrated in Fig. Simply put, there arent any atoms that are electronegative to. On the other hand, the ion pair 4-AT + NO 3 decomposes within 150-280☌ 30, 31 due to thermal decomposition of C 2 H 4 N 4 and HNO 3 fragments. Mesmer R E, Sweeton F H, Hitch B F and Baes C F 1976 In High-temperature high-pressure electrochemistry in aqueous solutions (eds) D D G Jones and R W Staehle (Houston, TX: National Association of Corrosion Engineers) pp 365–374 Hydrogen doesnt form ionic bonds because it has a very average electronegativity of 2.1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
